Top Grade Abaloparatide CAS 247062-33-5 For Reduced fracture rate and hypercalcemia incidence
Product Name: Abaloparatide
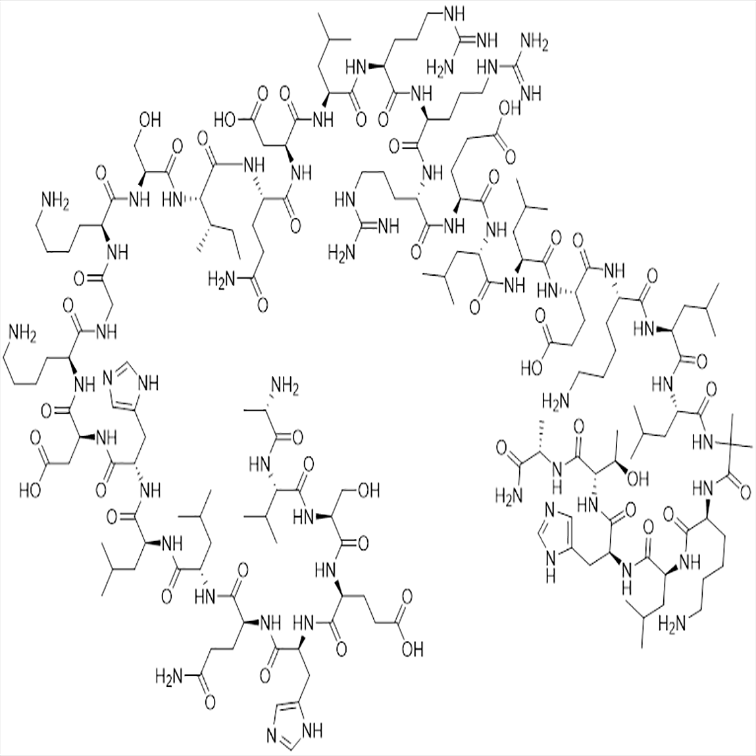
Sequence: H-Ala-Val-Ser-Glu-His-Gln-Leu-Leu-His-Asp-Lys-Gly-Lys-Ser-Ile-Gln-Asp-Leu-Arg-Arg-Arg-Glu-Leu-Leu-Glu-Lys-Leu-Leu-N-Me-Ala-Lys- Leu-His-Thr-Ala-NH2
Synonyms: Abaloparatide
CAS: 247062-33-5
MF: C174H300N56O49
MW: 3960.5896
EINECS:
Product Categories:
Mol File: 247062-33-5.mol
Abaloparatide, a selective activator of the parathyroid hormone receptor (PTH1 receptor), has been advanced into clinical development for the treatment of osteoporosis in postmenopausal women with a high risk of fracture due to its good osteogenic activity, made from abaloparatide.
Technical index Of Abaloparatide CAS 247062-33-5
Appearance: White powder
Purity(HPLC) ≥98.0%
Acetate content 5.0%~12.0%
Water content ≤8.0%
Peptide content≥80.0%
Packaging and transportation:
Low temperature, vacuum packed, accurate to mg upon request.
Function and Application Of Abaloparatide CAS 247062-33-5
Abaparatide, a peptide consisting of 34 amino acid residues, is a novel parathyroid hormone-related peptide (PTHrP) developed by RadiusHealth, which is a powerful and selective activator of the PTH-I receptor to increase bone mineral content, bone density, and bone strength and promote bone formation, and has been approved for marketing by the FDA on April Chemicalbook28, 2017. approved for marketing by the FDA on April 28, 2017 under the trade name Tymlos.Abapatide is administered subcutaneously for the treatment of osteoporosis in postmenopausal women who are at risk for fracture or who have failed to respond to other therapeutic agents, and is effective in reducing the rate of new vertebral and non-vertebral fractures. Abapatide is better able to reduce fracture rates and hypercalcemia rates compared to teriparatide.




Reviews
There are no reviews yet.