Hot Sale Albiglutide CAS 782500-75-8 treat for diabetes
Product Name: Albiglutide
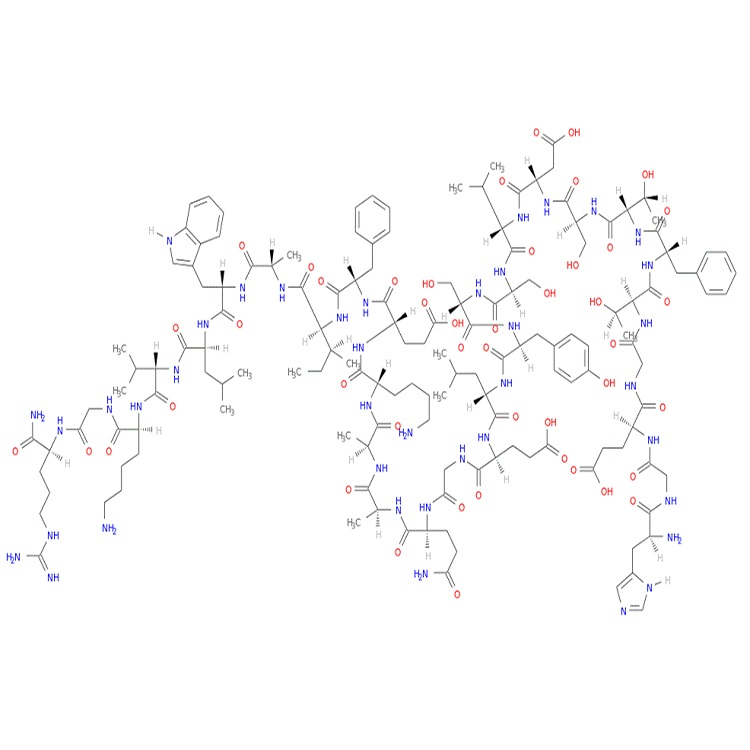
Sequence: H-His-Gly-Glu-Gly-Thr-Phe-Thr-Ser-Asp-Val-Ser-Ser-Tyr-Leu-Glu-Gly-Gln-Ala-Ala-Lys-Glu-Phe-Ile-Ala-Trp-Leu-Val-Lys-Gly-Arg-OH
Synonyms: albiglutide;Albugon;Naliglutide;7-36-Glucagon-like peptide I (8-glycine) (human), fusion protein with 7-36-glucagon-like peptide I (8-glycine) (human) fusion protein with serum;Gsk 716155;Unii-5E7U48495e;7-36-Glucagon-likepeptide I [8-glycine] (huMan), fusion protein with 7-36-glucagon-like peptide I[8-glycine] (huMan) fusion protein with seruM albuMin (huMan);Albiglutide Fragment
CAS: 782500-75-8
MF: C148H223N39O46
MW: 3284.58792
EINECS:
Product Categories: GLP
Mol File: 782500-75-8.mol
Albiglutide is FDA-approved for type 2 diabetes mellitus (DM) and prophylaxis for disorder of cardiovascular system as a result of type 2 diabetes. Its dosing involves titration to reduce gastrointestinal symptoms, such as nausea, diarrhea, and vomiting: initiate 0.6 mg daily subcutaneous injection for 1 week; increase to maintenance dose of 1.2 mg daily; and increase to 1.8 mg daily, as needed.
Liraglutide is also FDA-approved as Saxenda for obesity with a body mass index of 30kg/m2< or 27kg/m2 with the presence of at least 1 weight-related comorbid condition. Saxenda is initiated at 0.6 mg subcutaneous daily injection for 1 week, with weekly increments of 0.6 mg per day, until the maintenance dosage of 3 mg once daily is reached.3 If the patient misses a dose of liraglutide injection for more than 3 days, re-initiation at 0.6 mg is recommended to mitigate gastrointestinal (GI) symptoms.
Function and Application of Albiglutide CAS 782500-75-8
Back on April 15, 2014, the U.S. Food and Drug Administration (FDA) officially approved the diabetes drug Albiglutide for marketing. The drug is the second long-acting GLP-1 analogue approved to agonize the GLP-1 receptor and promote insulin secretion for the treatment of type 2 diabetes.






Reviews
There are no reviews yet.