Ganirelix Acetate CAS 123246-29-7 Gonadotropin-releasing hormone (GnRH) antagonists
Product Name: Ganirelix Acetate
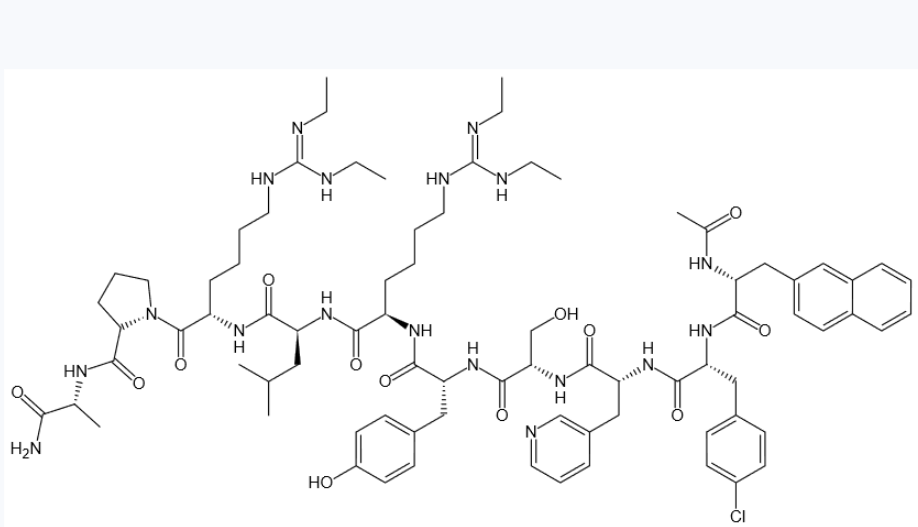
Synonyms: Ac-DNal-DCpa-DPal-Ser-Tyr-DHar(Et2)-Leu-Har(Et2)-Pro-DAla -NH2;Ganirelixum;ganirelix Acetate;GANIRELIX;Ganirelix Acetate USP/EP/BP
CAS: 123246-29-7
MF: C80H113ClN18O13
MW: 1570.34
EINECS:
Product Categories: hormones
Mol File: 123246-29-7.mol
Ganirelix acetate is a synthetic decapeptide compound similar to endogenous gonadotropin-releasing hormone (GnRH) (also known as luteinizing hormone-releasing hormone (LHRH)) and is an antagonist of GnRH that competitively antagonizes the gonadotropic GnRH receptor, thereby mutating the pathway and causing rapid and reversible inhibition of gonadotropin (luteinizing hormone LH and follicle stimulating hormone FSH) secretion. Ganirelix acetate inhibits LH secretion from the pituitary more significantly than FSH secretion, thereby reducing sex hormone production.
Function and Application of Ganirelix Acetate CAS 123246-29-7
Ganirelix acetate is an analog of GnRH with substitutions at residues 1, 2, 3, 6, 8, and 10. It is not a superagonist but, rather, is a synthetic decapeptide with high antagonist activity and the first GnRH antagonist to be marketed. It is approved for the suppression of LH surges in women who are undergoing ovarian hyperstimulation fertility treatment; LH surges normally promote ovulation. The goal of this drug is to significantly reduce the number of medication days necessary to suppress the LH surge, thereby maintaining eggs in the ovaries. In vitro fertilization (IVF) treatment cycles were historically initiated by the administration of leuprolide acetate to suppress the premature release of LH. This inhibits ovulation so that the eggs remain available for retrieval by a fertility specialist. For this purpose, leuprolide acetate usually is injected for as many as 26 days. Clinical studies have shown that ganirelix acetate can shut down the LH surge in only 5 days of treatment, that the suppression of LH is more pronounced than that of FSH, and that the shorter treatment time minimizes unpleasant side effects, such as hot flashes and headaches.
It is mainly used clinically in controlled ovarian stimulation protocols for women undergoing assisted reproductive technology to prevent premature luteinizing hormone peaks and for the treatment of fertility disorders caused by this cause Chemicalbook. The drug has fewer adverse effects, high pregnancy rates, and short treatment cycles, and has significant clinical advantages compared to similar drugs.






Reviews
There are no reviews yet.